By Brian Matovu, Biomedical Engineer, Innovator and Academician at Makerere University. Coordinator for CITE at Makerere University.
On 26th March 2026, Uganda’s Parliament passed the National Drug and Health Products Authority Bill, 2025 at its third reading; marking a decisive step toward transforming how health technologies are regulated in the country. This reform moves Uganda beyond a drug-centric regulatory model to a more comprehensive system that explicitly brings medical devices, diagnostics, and other health products into scope. In practical terms, this means that innovations which previously operated in regulatory grey areas such as locally developed maternal health diagnostics, CPAP devices, or surgical systems like gasless laparoscopy. These will now be subject to clearer, structured oversight across their lifecycle.
This shift is particularly important in addressing long-standing gaps in the system. For years, procurement and deployment environments in many low-resource settings have struggled with products entering the market with limited verification, sometimes carrying CE marks without accessible technical documentation or traceability to the original manufacturer. Under the new framework, stronger provisions for product registration, post-market surveillance, and recalls will help close this gap. For example, a diagnostic strip for early disease detection or a portable oxygen delivery system deployed in rural facilities will now require not just initial approval, but ongoing monitoring of safety and performance within the Ugandan context.
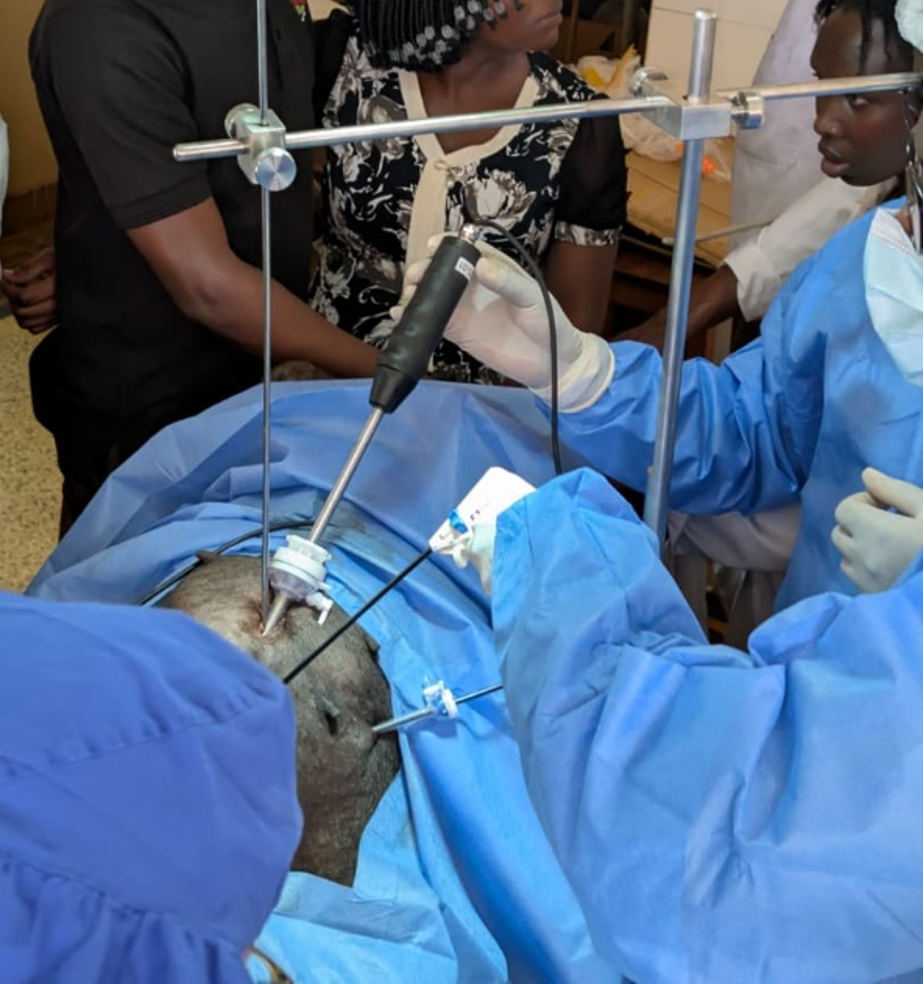
The KeySuite Laprascopic system undergoing wet lab testing at the College of Veterinary Medicine at Makerere University.

Assembly training of the LeVe CPAP system at Shishi International Limited which is an ISO 13485 certified manufacturing facility in Uganda.
The Bill also introduces the foundation for national testing and quality assurance infrastructure, which is critical for building trust in locally developed and imported technologies alike. Instead of relying solely on external laboratories or foreign regulatory decisions, Uganda can begin to verify performance domestically. This has direct implications for innovators working on solutions such as drug quality verification tools, point-of-care diagnostics, or reusable surgical instruments, where contextual validation (like local usage patterns, sterilization practices, and environmental conditions) is essential.
From a regional perspective, this positions Uganda among a growing group of African countries strengthening regulatory systems for health technologies. While South Africa’s SAHPRA has historically provided relatively mature oversight, many countries in the region are still developing clear pathways for medical devices and diagnostics. Uganda’s approach signals a move toward more harmonized, risk-based regulation, which could support cross-border recognition, regional manufacturing, and more efficient market access for innovators operating across East Africa and beyond.
Globally, the implications are even more compelling. High-income settings such as the European Union under the EU MDR or the United States through the FDA have long established rigorous frameworks for medical devices, including clinical evaluation, quality management systems, and post-market surveillance. Uganda’s reform reflects a growing recognition that similar principles adapted to resource-limited settings are essential for ensuring safety while enabling innovation. Importantly, this creates opportunities for reverse innovation and more inclusive regulatory science, where solutions are developed, tested, and validated in the contexts where they are most needed.
For institutions like Makerere University and initiatives such as CITE, this moment is both strategic and actionable. There is now a clearer mandate to support clinical investigations, generate local evidence, and contribute to the development of compliant manufacturing and quality systems. For example, a translational pathway can now be more deliberately defined from prototype development in the lab, to analytical validation using controlled samples, to clinical evaluation within Ugandan health facilities, and ultimately to regulatory approval and scale-up. This not only strengthens local capacity but also positions Uganda as a potential regional testbed for safe, scalable, and context-appropriate health technologies.
The real test, however, lies in implementation. The effectiveness of this reform will depend on how well regulators, academia, industry, and healthcare providers align to operationalize these provisions. If executed well, Uganda has the opportunity to move from being primarily a consumer of health technologies to becoming a contributor to global innovation—shaping how devices and diagnostics are designed, evaluated, and deployed in resource-constrained settings.
#MedicalDevices #GlobalHealth #Innovation #Uganda #RegulatoryScience #CITE #MakerereUniversity